Storing electricity has quietly become one of the biggest problems in the energy game. Solar panels only work when the sun shines and wind turbines rest on calm days, yet our homes, phones, and electric cars expect steady power around the clock. That is where a new kind of battery from China steps in with a surprising twist.
A team at the Dalian Institute of Chemical Physics has built the first functional hydrogen based battery that runs at room temperature using hydride ions as the carriers of charge. Their prototype, described in the journal Nature, shows that a concept long stuck on paper can actually power a real device.
Why a hydrogen battery matters for everyday energy use
Most people hear “hydrogen” and think of fuel cell cars or big industrial tanks. Here the element plays a different role since the battery uses tiny hydride ions, which are hydrogen atoms that carry an extra electron and move through a solid material. Researchers have eyed these ions for years because they are light, energetic, and in theory could store a lot of power in a small space.
Also Read: Treasury moves to end IRS union contracts
Until now, the big roadblock was the middle layer of the battery, called the electrolyte, which has to let hydride ions flow while staying stable and safe. Earlier candidates either moved ions too slowly, reacted with the electrodes, or broke down when heated.
In practice that meant hydride batteries stayed stuck in the lab as elegant theories but not something that could help with your electric bill or keep solar power flowing after sunset.
How the Chinese team made hydride ions work
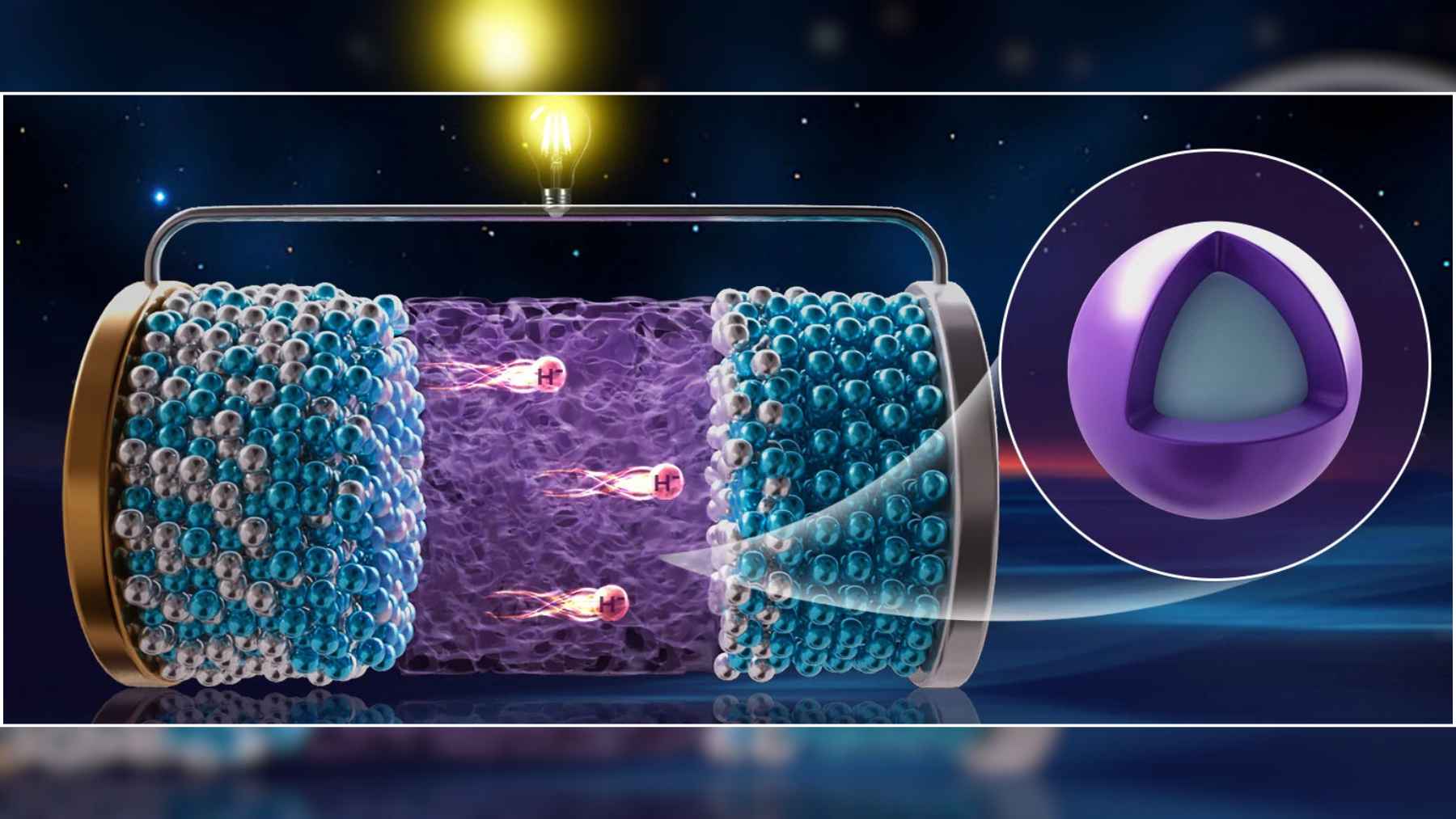
To solve that problem, the group led by Ping Chen designed a new solid material with a so called core shell structure. At its center sits cerium hydride, which lets hydride ions move quickly, while the surface is wrapped in a thin layer of barium hydride that keeps the material stable and compatible with the rest of the battery.
The result is a composite known as 3CeH3@BaH2 that conducts hydride ions at room temperature and, when warmed above about 60 degrees Celsius, behaves like a super fast ionic conductor. In simple terms, it acts like a highway for hydrogen based charge inside a fully solid device. That is new.
With this electrolyte in hand, the researchers assembled a cell with a positive electrode made from a sodium aluminum hydride compound and a negative electrode made from cerium hydride, both already familiar in experimental hydrogen storage work. Sandwiched between them, the new material allows hydride ions to shuttle back and forth as the battery charges and discharges.
What the first tests reveal about performance
In early tests, the positive electrode delivered an initial discharge capacity close to 984 milliampere hours per gram at room temperature, a figure that expresses how much charge the material can hold for its weight. After twenty charge and discharge cycles, it still provided around 402 milliampere hours per gram, so there is some loss but the system remains clearly active.
The team also stacked several cells together and reached an operating voltage of about 1.9 volts. That may sound modest compared with the battery pack in an electric car, yet it was enough to light up a yellow LED lamp, moving the concept firmly out of computer models and into a working device you can see with your own eyes.
One important claim from the researchers is that using hydrogen as the charge carrier should avoid the metal dendrites that sometimes grow in lithium batteries and can cause short circuits or fires.
In their words, “the use of hydrogen as a charge carrier avoids the formation of dendrites and opens new routes for clean energy storage and conversion”. That suggests potential gains in safety and long term durability if the idea scales up.
Still a long road from lab bench to power grid
Can this hydrogen battery compete with the lithium cells inside your phone or parked electric car. Not yet. The prototype has only been cycled a few dozen times, and commercial products usually need to last for hundreds or thousands of cycles while handling heat, cold, and daily abuse.
Engineers will also have to figure out how to manufacture the new electrolyte in large volumes, connect many cells safely, and keep costs under control. That is especially important if this technology is ever used to back up large solar farms during that sticky summer heat when air conditioners are running full blast.
At the end of the day, what the current work really offers is proof that hydride batteries are more than a thought experiment.
Even so, the step from theory to a working solid state hydrogen battery is significant. If future versions can hold their capacity for longer and remain stable in real world conditions, they could join lithium, sodium, and other chemistries in a more diverse and resilient energy storage mix.
The main study has been published in the journal Nature.